- Blog
- Cursive fonts tattoos
- Minecraft download unblocked 1-5-2
- Periodic table is molar mass or amu
- Spectrum basic tv channels list
- Thompson center contender serial number dates
- Sight words list kindergarten
- Rpg maker vx ace tilesets template
- Good keno numbers
- Mickey-s castle of illusion ps3 rom
- App background eraser --
- Xbox 360 afterglow controller software
- Colorcross crossword answers
- United states doppler weather radar map
- Clock on windows 10 desktop
- Cool house ideas terraria
- Dsm 5 criteria for diagnosing ptsd
- Northern california fire map
- Gacha life games free
- Default dance copy and paste text art
- Save wizard license key download crack
- Rainmeter skins clock time zone download
- Blog
- Cursive fonts tattoos
- Minecraft download unblocked 1-5-2
- Periodic table is molar mass or amu
- Spectrum basic tv channels list
- Thompson center contender serial number dates
- Sight words list kindergarten
- Rpg maker vx ace tilesets template
- Good keno numbers
- Mickey-s castle of illusion ps3 rom
- App background eraser --
- Xbox 360 afterglow controller software
- Colorcross crossword answers
- United states doppler weather radar map
- Clock on windows 10 desktop
- Cool house ideas terraria
- Dsm 5 criteria for diagnosing ptsd
- Northern california fire map
- Gacha life games free
- Default dance copy and paste text art
- Save wizard license key download crack
- Rainmeter skins clock time zone download
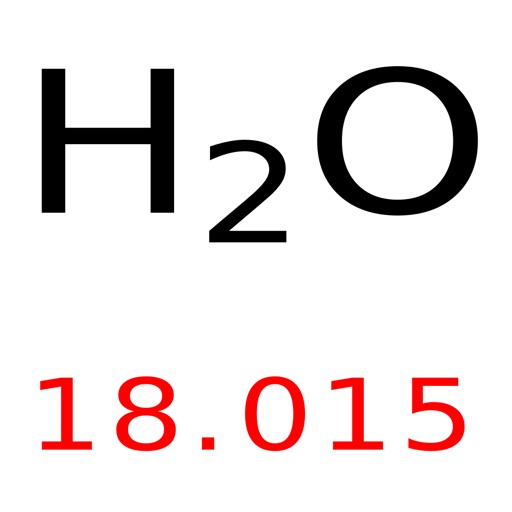
(in other words we reduced 100% to decimal form 1. The periodic table lists the atomic mass of carbon as 12. We will let 6Li = x and 7 Li = 1-x we use 1 – x instead of 100 – x because the small number is easier to work with. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (NO 2 (amu)) Count i Weight i.
:max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
Then, lookup atomic weights for each element in periodic table: N: 14.0067, O: 15.9994. Since I don’t know what the percentage are, I will have to use variables.ġ00% of Lithium is determined by these two naturally occurring isotopes. First, compute the number of each atom in NO 2 (amu): N: 1, O: 2. Determine the percent abundance of each isotope.Īw = + + Ħ.94 = + The atomic mass of lithium is 6.94, the naturally occurring isotopes are 6Li = 6.015121 amu, and 7Li = 7.016003 amu. What are the percent abundances of the isotopes? Since the overall atomic weight for copper is not given in the problem, you must look it up in the periodic table to work this solution. If you look in the periodic table you will be able to check that our answer is correct!ģVerify that the atomic mass of magnesium is 24.31, given the followingĪtomic mass= + + ĭetermining the percent abundance of each isotope from atomic mass.Ĭopper exists as two isotopes: 63Cu (62.9298 amu) and 65Cu (64.9278 amu). 10.81amu so, the atomic weight of B = 10.81amu
